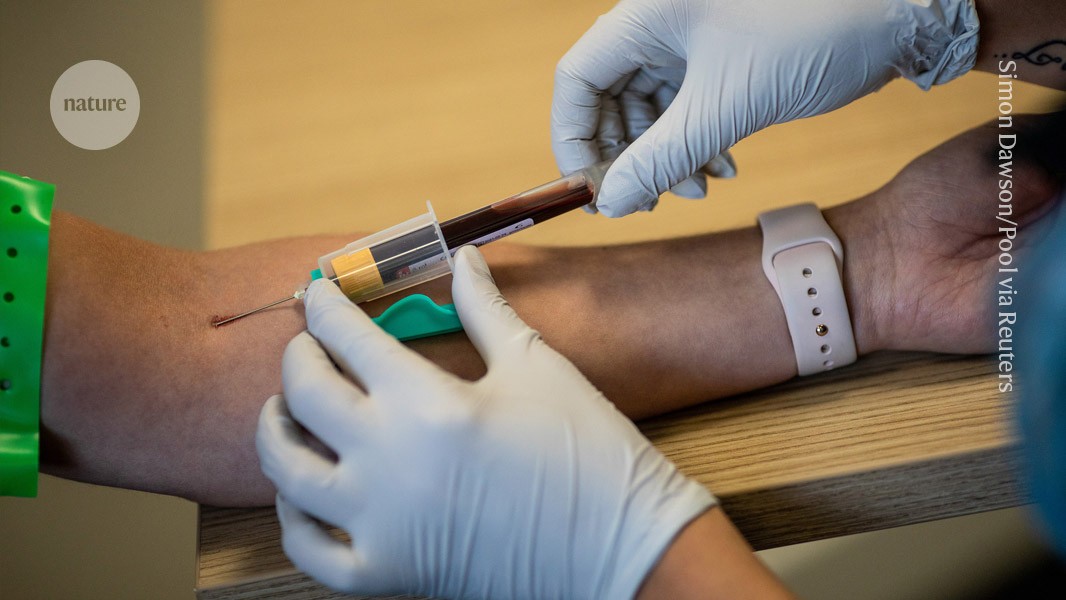
"The test, called Galleri, is one of around 40 such multi-cancer early detection (MCED) tests that are either in development or already on sale. But very few have been through randomized controlled trials (RCTs) - which are considered the gold standard of testing - and none has received approval from regulators."
"Last month, the developers of the Galleri test, biotechnology company Grail in Menlo Park, California, released some details from the first RCT of an MCED test. The trial, run in collaboration with the UK National Health Service (NHS), aimed to find out whether Galleri can improve outcomes by reducing the number of cancers detected at advanced stages, when used alongside existing screening programmes - but the results indicate that the test did not meet this goal."
"The science that powers these tests is not new: researchers have long known that markers of cancer, including fragments of DNA shed by cancer cells, can show up in blood, saliva and urine. And 'liquid biopsy' tests, based on similar technologies to those that underpin MCED tests, are already widely used by physicians to monitor cancer progression and choose the best treatments."
Multi-cancer early detection (MCED) tests, including Galleri, detect cancer markers in blood to identify over 50 cancer types. Approximately 40 such tests are in development or available, yet few have undergone randomized controlled trials and none has received regulatory approval. Galleri's first RCT, conducted with the UK NHS, failed to demonstrate improved outcomes by reducing advanced-stage cancer detection when used alongside existing screening programs. While liquid biopsy technology underlying these tests is established and used clinically to monitor cancer progression, detecting early-stage cancer remains challenging due to minimal tumor DNA presence in blood. Researchers debate whether MCED tests could revolutionize cancer detection, particularly for cancers lacking screening tools, against concerns about low detection rates and false diagnosis consequences.
Read at Nature
Unable to calculate read time
Collection
[
|
...
]