fromwww.theguardian.com
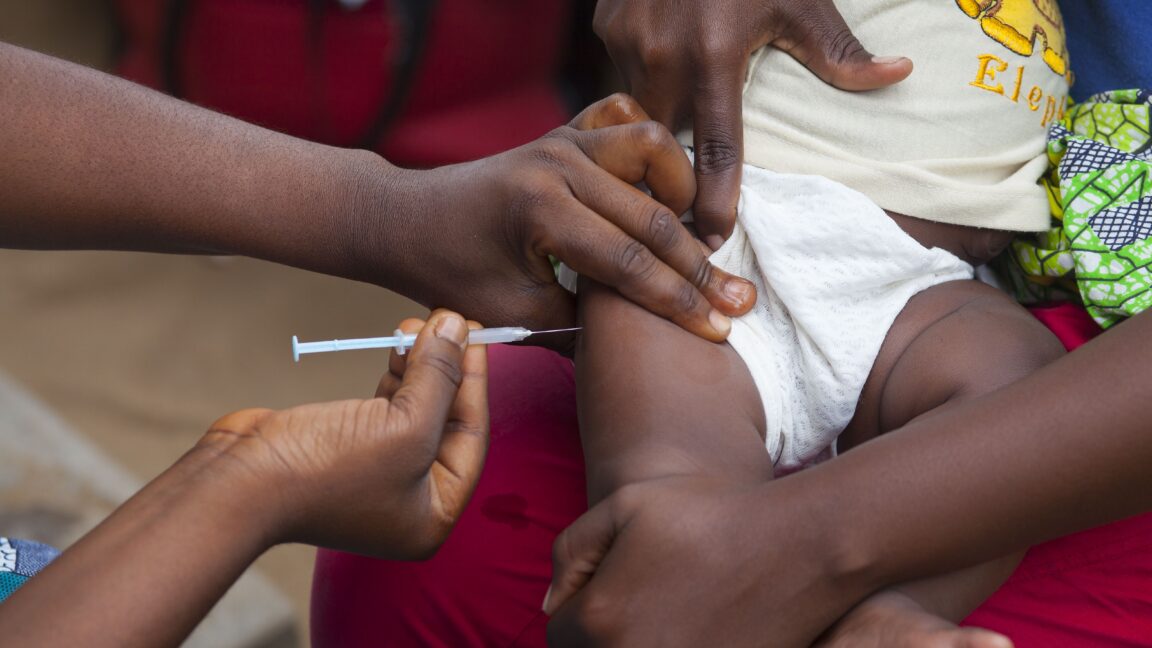
1 month agoExperts fear unethical' vaccine trial in Africa is prototype' for US studies under RFK Jr
We are fearful that this is a prototype for other studies. The US could fund global studies with the similar ethical concerns as the Tuskegee experiment five or 10 or 100 times a year. It could be extraordinarily deadly.
Public health