#drug-pricing-deal
#drug-pricing-deal
[ follow ]
Intellectual property law
fromIPWatchdog.com | Patents & Intellectual Property Law
4 years agoMossoff Policy Memo for Hudson Institute Calls for Transparency from I-MAK on Data Used in Drug Pricing Debate
Discrepancies exist between patent data reported by I-MAK and official sources, raising concerns about the reliability of data in pharmaceutical patent debates.
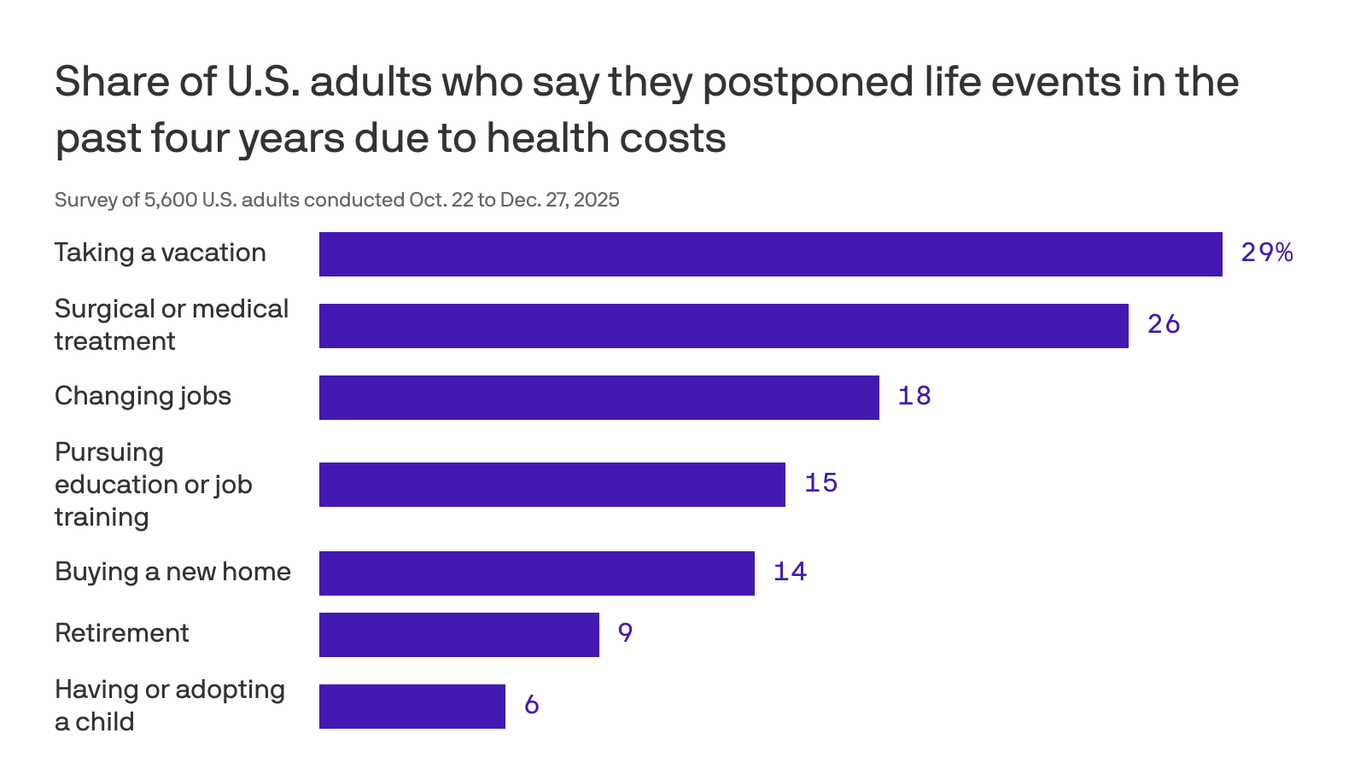
Retirement
from24/7 Wall St.
2 weeks agoThe $2,000 Drug Cap Is Saving Medicare Retirees Over $1,500 a Year Right Now
Medicare premiums in 2026 are based on 2024 income, creating unexpected surcharges for those with one-time income spikes, while Part B premiums exceed $200 monthly and a new rule allows tax-free distributions for long-term care insurance.
Intellectual property law
fromIPWatchdog.com | Patents & Intellectual Property Law
1 week agoU.S. Government Says SCOTUS Should Skip Pharma Companies' Challenge to Medicare Negotiation Program
The Medicare Drug Price Negotiation Program does not constitute a physical taking under the Takings Clause, allowing companies to retain property rights.
US news
fromwww.npr.org
3 weeks agoMedicare Advantage 'dark money' group tries to win higher payments for insurers
A dark money group orchestrated a coordinated campaign with 83% identical comments on federal Medicare Advantage reimbursement proposals, masking industry opposition as grassroots advocacy.
Public health
fromwww.theguardian.com
3 weeks agoPatients face long journeys for medicines as pharmacies cut weekend hours
One in six English pharmacies have reduced weekend hours since 2022, causing over 20% loss of weekend opening hours and forcing patients to travel long distances or seek emergency care.
#prescription-drug-pricing
US Elections
fromwww.mediaite.com
3 weeks agoTrump Uses Numerically Possible Rx Price Percentages
Trump claimed prescription drug price reductions of 70-90% in a local interview, abandoning his previous false claims of reductions exceeding 100%, though his statements about U.S. drug prices being lowest globally remain unsubstantiated.
Alternative medicine
fromNatural Health News
3 months agoTrump administration secures historic Medicare drug price cuts, challenging Big Pharma's grip
Medicare negotiated 44% price cuts on 15 expensive drugs, projecting $12 billion in savings and establishing the first federal government direct drug price negotiation under the 2022 Inflation Reduction Act.
fromMail Online
3 weeks agoOutrage as cancer-fighting drug in US patent echoes hidden CIA file
According to the patent, a specific crystalline form of the drug known as polymorph C may be more effective than other versions because it is absorbed more efficiently by the body. The patent also notes that laboratory studies showed the drug reduced tumor growth and helped mice with brain tumors live longer, prompting early clinical trials to test whether the treatment is safe and effective in humans.
Cancer
Public health
fromLGBTQ Nation
1 month ago17 states consider cutting assistance for HIV meds as prices increase - LGBTQ Nation
Seventeen states and D.C. have cut AIDS Drug Assistance Program costs while federal funding stagnates, threatening medication access for low-income HIV patients and risking public health crises.
Healthcare
fromHarvard Gazette
3 weeks agoFederal drug price reforms are working, finds study - Harvard Gazette
Medicare beneficiaries reported skipping fewer medications due to cost after the Inflation Reduction Act's prescription drug provisions took effect in January 2024, with greatest improvements among those with multiple chronic conditions.
Healthcare
fromMedCity News
3 weeks agoIt Took Years for Congress to Enact PBM Transparency, Delinking. What About Vertical Integration? - MedCity News
Congress passed initial PBM reforms in February 2026, but advocates seek stronger action against vertical integration between PBMs, insurers, and pharmacies through proposed legislation like the Break Up Big Medicine bill.
fromIPWatchdog.com | Patents & Intellectual Property Law
2 months agoThe Federal Government's Drug Price Negotiation Program Would Likely Violate Its Own Antitrust Laws
The IRA requires drugmakers to sell selected patented drugs to the government for its Medicare Parts B & D programs at a stipulated "maximum fair price". If they don't agree to these prices, then they face tax penalties on sales of the drug exceeding their profits from it, or the exclusion of all their drugs from Medicare and Medicaid purchases. This would foreclose access to up to 160 million patients, accounting for around 40% of US prescription drug spending or 20% of global prescription drug spending.
US politics
fromwww.npr.org
1 month agoHospitals are posting prices for patients. It's mostly industry using the data
The idea echoes a policy implemented during his first term, when Trump suggested that requiring hospitals to post their charges online could ease one of the most common gripes about the health care system the lack of upfront prices. To anyone who's gotten a bill three months after treatment only to find mysterious charges, the idea seemed intuitive. "You're able to go online and compare all of the hospitals and the doctors and the prices,"
US news
Public health
fromkffhealthnews.org
2 months agoMedical bills can be vexing and perplexing. Here's important advice for patients
High and confusing medical bills affect insured and uninsured due to coverage gaps, unexpected plan restrictions, billing practices, and complex retroactive policies like COBRA.
Healthcare
fromFast Company
1 month agoResponsible compounding could close the innovation gap
Compounding can responsibly accelerate patient access to needed therapies when grounded in rigorous data, filling genuine clinical gaps while pursuing FDA approval, particularly in underserved areas like women's health.
Healthcare
fromBusiness Insider
1 month agoWaiting for Medicare and skipping prescriptions: Meet the Americans on the brink of losing health insurance
Expiration of enhanced ACA subsidies on December 31 caused monthly premiums to spike hundreds of dollars, forcing over 1 million Americans to cancel marketplace plans between early 2025 and 2026.
Healthcare
fromwww.npr.org
1 month agoCan't get a prescription renewed? Here's how to cope with prior authorizations
Insurance prior authorization requirements expire even for patients already taking prescribed medications, forcing repeated approval processes and potentially interrupting effective treatments.
fromFlowingData
2 months agoWhere generic medication comes from
When generic drug manufacturers have issues like contamination, it is difficult for those who take the medications to know if they are affected. There is no standardized way to look up the data for where the pills in your bottle came from. ProPublica made an app that makes the lookup more straightforward. Even though generic drugs make up 90% of prescriptions dispensed in the U.S., the FDA only provides piecemeal information about them.
Public health
fromMedCity News
1 month agoWhy HHS Scrapping Its 340B Rebate Program Is a Win For Providers - MedCity News
The 340B program allows hospitals to buy outpatient drugs at steep discounts, with the purported purpose of helping them fund care for low-income and uninsured patients. The now-axed rebate model would have invited drugmakers to participate voluntarily in a rebate-based discount system. Basically, instead of the provider receiving a discount upfront at purchase, the 340B discount would be applied after purchase via rebate - and subject to tedious data submission requirements.
Public health
fromwww.scientificamerican.com
1 month agoWhat if most medications were sold over-the-counter?
The U.S. Food and Drug Administration is reportedly mulling whether more prescription drugs should be sold over the counter (OTC) at pharmacies. In an interview on Wednesday, FDA commissioner Martin Makary told CNBC that everything should be over the counter except drugs that are deemed unsafe or addictive or that require clinical monitoring. Makary said the agency is reviewing how it decides which drugs can be sold with or without a prescription from a health care practitioner.
Healthcare
fromMedCity News
1 month agoEmployers Celebrated PBM Reform. The Reality Is More Complicated - MedCity News
For years, Congress has signaled that it wants to crack down on Pharmacy Benefit Managers, the middle men that have come under fire for their vertical integration with insurers and their role in spiking drug costs. This week, it finally happened via the Consolidated Appropriations Act of 2026, promptingemployer groups including the Purchaser Business Group on Health (PBGH) and the ERISA Industry Committee to cheer its passage.
Healthcare
[ Load more ]